“Eat your green veggies!” We’ve been telling blood donors things like that for decades, but is it really helpful? Dr. Jed Gorlin explains the recent, troubling data on iron and blood donors.

Dr. Jed Gorlin
So What do we Do?
This is an extremely important discussion, as blood centers everywhere are struggling with decreased collections. It is tempting to depend on donors in certain “easy to collect” groups, and those include donors at high risk outlined above! So, what to do? Dr. Gorlin has been heavily involved in these discussions, and he brings a unique perspective to this interview. He describes the relevant studies (linked in the “Further Reading” section below), and walks us through the steps that blood collectors (and donors) should take right away.

Dr. Jed Gorlin
So What do we Do?
This is an extremely important discussion, as blood centers everywhere are struggling with decreased collections. It is tempting to depend on donors in certain “easy to collect” groups, and those include donors at high risk outlined above! So, what to do? Dr. Gorlin has been heavily involved in these discussions, and he brings a unique perspective to this interview. He describes the relevant studies (linked in the “Further Reading” section below), and walks us through the steps that blood collectors (and donors) should take right away.
About My Guest:
Dr. Jed Gorlin received his BS from Stanford and his MD from Yale. He is board-certified in Pediatrics and Blood Banking/Transfusion Medicine. He is Medical Director and Vice President, Quality and Regulatory Affairs at Innovative Blood Resources. Jed’s duties include Memorial Blood Centers in Minnesota, Nebraska Community Blood Bank, the Community Blood Center of Greater Kansas City in Kansas City, MO. He has served in various leadership roles with AABB through his more than 20 years in Transfusion Medicine, including Chair of the Standards Committee for the 20th and 21st editions of AABB Standards, and as an AABB board member. He is a chapter contributor to 21 published books and anthologies, and author or co-author of over 120 published papers and abstracts.
The images below are generously provided by Dr. Gorlin.
References Mentioned During this Episode:
- RISE Enrollment Data: Cable RG, Glynn SA, Kiss JE, et al. Iron Deficiency in Blood Donors: Analysis of Enrollment Data from the REDS-II Donor Iron Status Evaluation (RISE) Study. Transfusion. 2011;51(3):511-522.
- RISE Results: Cable RG, Glynn SA, Kiss JE, et al. Iron Deficiency in Blood Donors: The REDS-II Donor Iron Status Evaluation (RISE) Study. Transfusion. 2012;52(4):702-711.
- Study Showing Diet Alone is Insufficient for Frequent Blood Donors: Rigas A, Sorensen C, Pedersen O, et al. Predictors of iron levels in 14,737 Danish blood donors: results from the Danish blood study. Transfusion 2014;54:789-96
- HEIRS: Kiss JE, Brambilla D, Glynn SA et al for the National Heart, Lung, and Blood Institute (NHLBI) Recipient Epidemiology and Donor Evaluation Study–III (REDS-III). Oral iron supplementation after blood donation: A randomized clinical trial. JAMA. 2015;313(6):575-583
- HEIRS Analysis of Effect: Cable RG, Brambilla D, Glynn SA et al for the National Heart, Lung, and Blood Institute Recipient Epidemiology and Donor Evaluation Study-III (REDS-III). Effect of iron supplementation on iron stores and total body iron after whole blood donation. Transfusion, 56: 2005–2012.
- STRIDE: Mast AE, Bialkowski W, Bryant BJ et al. A randomized, blinded, placebo-controlled trial of education and iron supplementation for mitigation of iron deficiency in regular blood donors. Transfusion 2016;56:1588–1597.
- FDA Blood Products Advisory Committee: Discussion of iron management in blood donors, from November 2016
- FDA Blood Products Advisory Committee: Discussion of iron deficiency in teenage donors from November 2016
- AABB: Summary of BPAC Meeting (includes preliminary data on CHILL results at end of “Topic II” discussion).
- AABB: Public statement on iron management in blood donors, May 2017

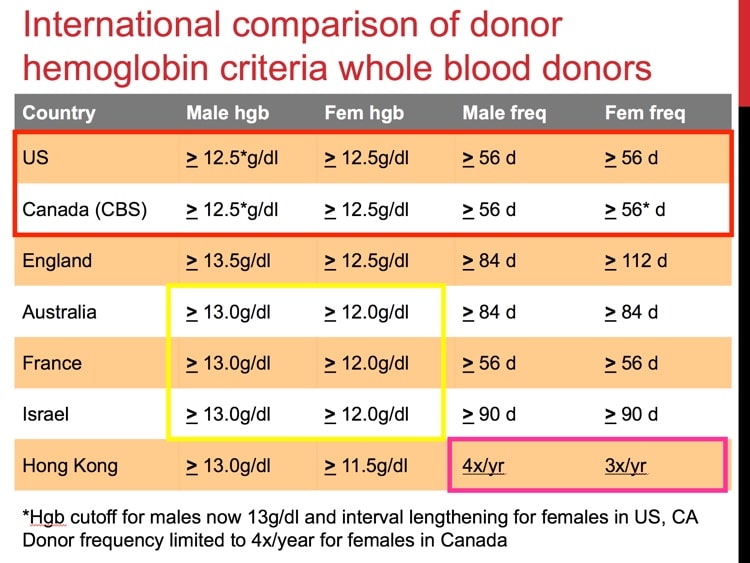
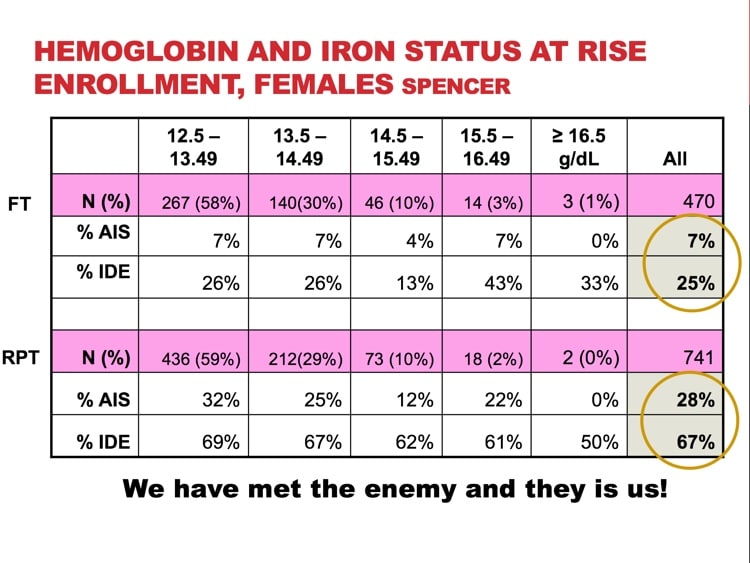
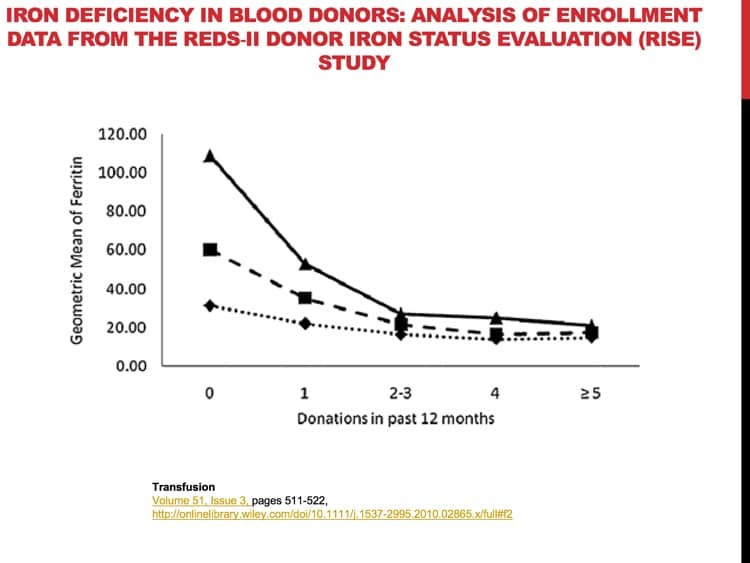



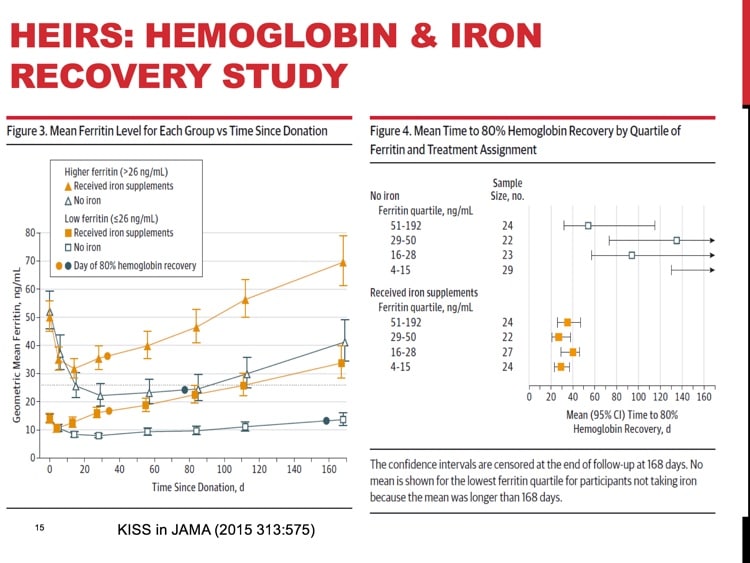
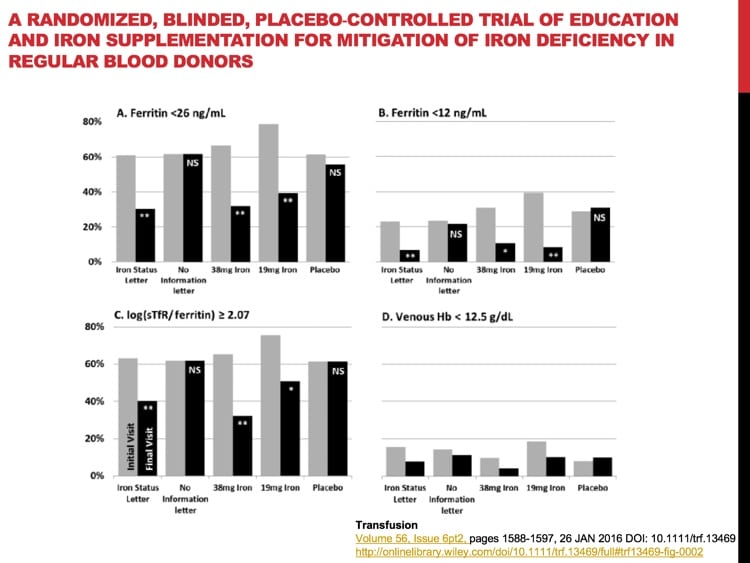










So what is the desired Iron/Ferrittin levels the donor should have in their blood?
Mariam, donors who are NOT iron-depleted, according to the HEIRS and STRIDE studies mentioned, were those with ferritin levels above 26 ng/mL.
-Joe